Dangerous spattering of strong acid or base can be avoided if the concentrated acid or base is slowly added to water, so that the heat liberated is largely dissipated by the water. The lattice energy of Calcium chloride CaCl 2 is-2195. As defined in Equation 1 1, the lattice energy is positive, because energy is always required to separate the ions. If water is added to a concentrated solution of sulfuric acid (which is 98% H 2SO 4 and 2% H 2O) or sodium hydroxide, the heat released by the large negative Δ H can cause the solution to boil. It can also be calculated from the electrostatic consideration of its crystal structure. This phenomenon is particularly relevant for strong acids and bases, which are often sold or stored as concentrated aqueous solutions. Use the Kapustinskii equation to estimate the lattice energy for CaCl2.  
Lattice energy CaCl:Ca2+ (g)+2Cl (g)CaCl2 (s) latt HkJmol1 B. If the initial dissolution process is exothermic (Δ H < 0), then the dilution process is also exothermic. Use the values below to calculate the lattice of enthalpy of CaCl2. The Δ H soln values given previously and in Table 8.2.2 for example, were obtained by measuring the enthalpy changes at various concentrations and extrapolating the data to infinite dilution.īecause Δ H soln depends on the concentration of the solute, diluting a solution can produce a change in enthalpy. The amount of heat released or absorbed when a substance is dissolved is not a constant it depends on the final concentration of the solute. Because of the mass of white sodium acetate that has crystallized, the metal disc is no longer visible. For the process to be spontaneous, G (dissolution)<0. G H-TS for completely understanding the solubility trends. 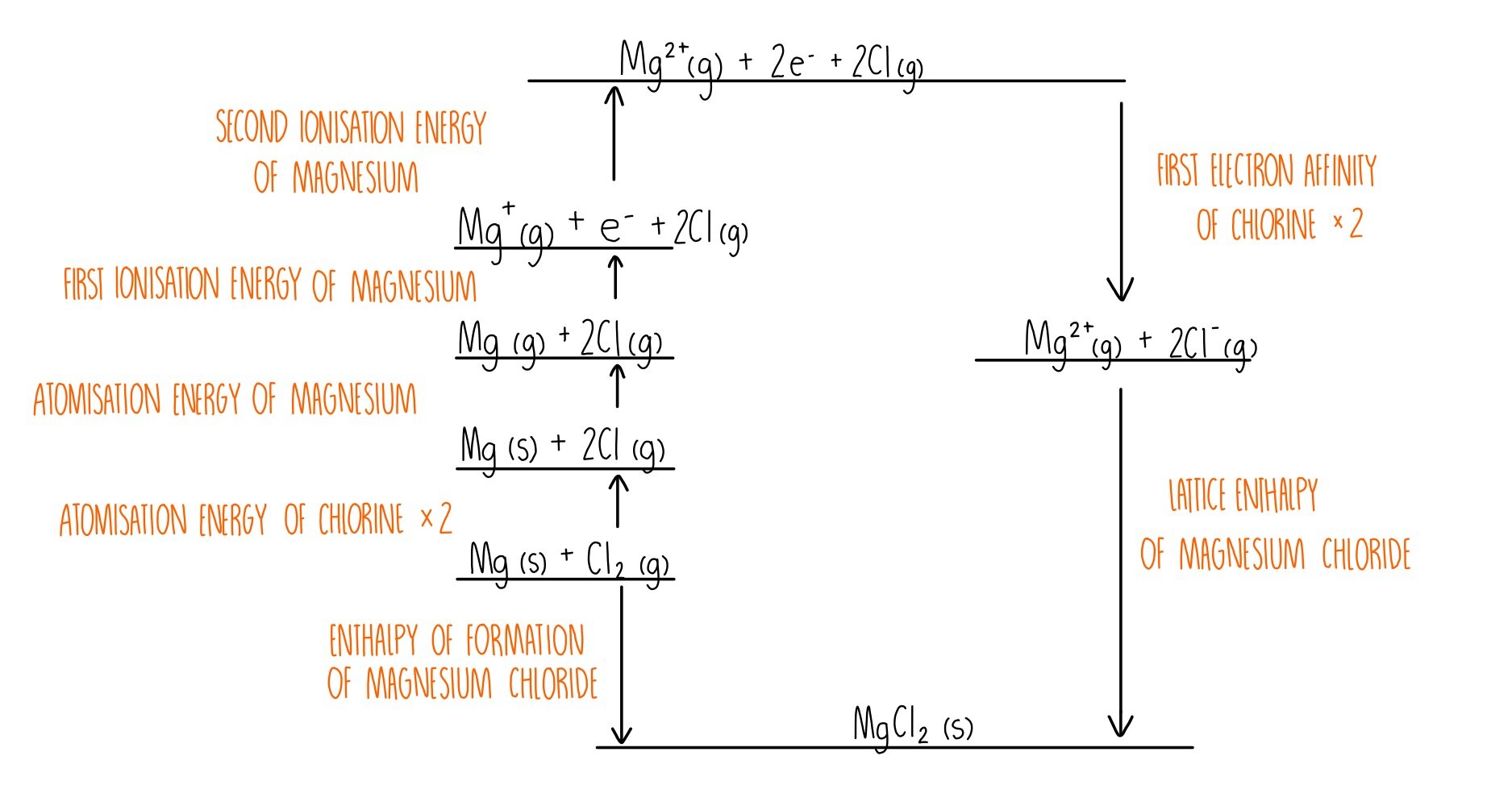
So being a thermodynamic concept, we will have to turn to Gibbs free energy i.e. After the hot pack has been agitated, the sodium acetate crystallizes (right) to release heat. The thing you have to understand is that the enthalpy of dissolution is actually the sum of lattice enthalpy and hydration enthalpy. Because the sodium acetate is in solution, you can see the metal disc inside the pack. When the pack is agitated, sodium acetate trihydrate crystallizes, and heat is evolved:įigure 9.5.1 An Instant Hot Pack Based on the Crystallization of Sodium Acetate The hot pack is at room temperature prior to agitation (left). With cooling, an unstable supersaturated solution containing excess solute is formed. The science, which deals with crystals properties is. The lattice energy is usually given in kilojules per mole (kJ/mol). (ii) Use the data in the following table and from the data booklet to construct the Born-Haber cycle for calcium chloride, CaCl2, and determine the lattice enthalpy of CaCl2(s). The lattice energy is the total potential energy of the crystal. Based on the lattice energies, would you expect the NaCl or the KF distance to be longer The substances NaF and CaO are isoelectronic (have the same number of valence electrons). (a) The lattice energies of NaCl and KF are given in Table 8.1. At high temperatures, sodium acetate forms a highly concentrated aqueous solution. Equivalently, lattice energy can be defined as the amount of work (energy) that is released during creation of crystal lattice from ions separated to infinity. The only difference between the two is the distance that separates cations and anions. If the salt is CaCl 2, heat is released to produce a solution with a temperature of about 90☌ hence the product is an “instant hot compress.” If the salt is NH 4NO 3, heat is absorbed when it dissolves, and the temperature drops to about 0° for an “instant cold pack.”Ī similar product based on the precipitation of sodium acetate, not its dissolution, is marketed as a reusable hand warmer ( Figure 9.5.1). When the pack is twisted or struck sharply, the inner plastic bag of water ruptures, and the salt dissolves in the water. Both types consist of a plastic bag that contains about 100 mL of water plus a dry chemical (40 g of CaCl 2 or 30 g of NH 4NO 3) in a separate plastic pouch. Single-use versions of these products are based on the dissolution of either calcium chloride (CaCl 2, Δ H soln = −81.3 kJ/mol) or ammonium nitrate (NH 4NO 3, Δ H soln = +25.7 kJ/mol). Calculate the lattice enthalpy of CaCl2, given that the enthalpy of (i) sublimation of Ca is 121 kJ/mol (ii) dissociation of Cl2 to Cl is 242. Substances with large positive or negative enthalpies of solution have commercial applications as instant cold or hot packs. /SRP: Calcium gluconate has replaced many therapeudic indications for calcium chloride. Table 9.5.1 Enthalpies of Solution at 25☌ of Selected Ionic Compounds in Water (in kJ/mol) We begin with the elements in their most common states, Cs( s) and F 2( g).\) \): The Born-Haber cycle shows the relative energies of each step involved in the formation of an ionic solid from the necessary elements in their reference states.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
